Renowned Speakers

Jun Yang
Western University, Canada Canada

Sang Youl Kim
Korea Advanced Institute of Science and Technology South Korea

Sevinc Ilkar Erdagi
Kocaeli University Turkey

Adam Quintana
University of New Mexico USA

Byung Gil Min
Kumoh National Institute of Technology South Korea

Ricardo Marques e Silva
McMaster University Canada

Jordan Milne
McMaster University Canada

Shih-Chieh Hsiao
National Cheng Kung University Canada
Recommended Global Chemistry Webinars & Conferences
Europe & UK
Asia Pacific & Middle East
Canada
Polymer Chemistry 2023
About Polymer Chemistry 2023
Polymer Chemistry 2023 International Conference is an attempt to explore the various ways to utilize natural resources for betterment of the future, promising a better tomorrow for the progeny and a better vision for the springing research. Polymer Science and Polymer Chemistry are anticipated to be one the best scientific conferences in Rome. The scientific sessions of this Polymer Chemistry 2023 has been designed on vivacious topics such as Polymer Recycle and Polymer Waste Management, Biodegradable Plastics Applications, Green Composites in Biopolymers. Polymer Waste conferences are consisting of well-organized scientific program and effervescent speeches by the expertise.
Polymer Chemistry aims to bring together scientists, researchers, and practitioners in order to discuss and share cutting-edge development in the field. Polymer Chemistry provides an ideal platform and opportunity for all the young researchers to connect with eminent Scientists and Industrialists. The Conference is a premier enlightening and networking for all industry stakeholders, policy makers, investors, industry and research community to exchange experiences and challenges related to development and scaling up in the field of chemical engineering, smart materials, biopolymers and materials science.
Session & Tracks
Track 1: Polymer Chemistry
Polymer scientific experts' examination huge, complex atoms (polymers) that are upgrade from numerous more modest (sometimes repeating) units. They concentrate how the more modest structure blocks (monomers) fuse, and make helpful materials with explicit attributes by controlling the sub-atomic construction of the monomers/polymers utilized, the synthesis of the monomer/polymer solidification, and applying substance and preparing procedures that can, generally, influence the properties of the end result. Polymer scientific experts are diverse inside the science local area in light of the fact that their comprehension of the connection among construction and property ranges from the sub-atomic scale to the perceptible scale.
Recommended Conferences:
Materials Chemistry 2023 | World Chemistry 2023 | Euro Biosensors 2023 | Euro Chemistry 2023 | Organic Chemistry 2023 | Environmental Chemistry 2023 | Physical Chemistry 2023 | Pharmaceutical Chemistry 2023 | Electrochemistry 2023 | Separation Techniques 2023
Polymer Companies in Europe
Leibniz Institute of Polymer Research Dresden | Postnova Analytics GmbH | Addlink software cientifico | CK Polymers | Advanced Technologies Center ATC | Magpie Polymers | Living Cell Technologies | Micro-Bio-Polymer Company | Evolve Polymers Limited | Meric Ltd | Quick step Technologies | VSPC co.LTD | Warsash scientific | AQUANOVA AG | Gyros | Membracon Process Separations | EPC Engineering Consulting GmbH | VIP Polymers Ltd | nDure Technologies | Ingenia Polymers Corp. | Quick step Technologies | Gioco Solutions Sr l CETCO Energy Services | Dr. Jens Naehring | POLYMERS & COLORS S.A. | NOXON AB
Related Societies:
Royal Australian Chemical Institute | Brazilian Chemical Society | Asian Polymer Association | Society of Chemical Industry | Society of Chemical Manufacturers and Affiliates | Royal Society of Chemistry | European Association for Chemical and Molecular Sciences | European Federation of Chemical Engineering | Royal Institute of Chemistry | ACS Division of Polymer Chemistry | American Chemical Society Division of Polymeric Materials | Canadian Society of Clinical Chemists
Track 2: Polymer Synthesis
Polymer researchers have been made an alternate cultivating system research in the advancement of biodegradable polymers, which could discover colossal applications in the space of clinical science. Today, extraordinary biopolymers have been arranged and used in various biomedical applications. In spite of the obvious multiplication of biopolymers in clinical science, the science and innovation of biopolymers is as yet in its beginning phases of advancement. Huge window exists and will stay to exist for the infiltration of biopolymers in each aspect of clinical science through escalated innovative work. Accordingly, this part tends to different polymerization strategies and methods utilized for the arrangement of biopolymers. The accentuation is on the properties of biopolymers, engineered conventions, and their biomedical applications. To make the helpful biomedical gadgets from the polymers to fulfill the needs of clinical science, different handling procedures utilized for the improvement of gadgets have been examined.
Recommended Conferences:
Materials Chemistry 2023 | World Chemistry 2023 | Euro Biosensors 2023 | Euro Chemistry 2023 | Organic Chemistry 2023 | Environmental Chemistry 2023 | Physical Chemistry 2023 | Pharmaceutical Chemistry 2023 | Electrochemistry 2023 | Separation Techniques 2023
Polymer Companies in Europe
Leibniz Institute of Polymer Research Dresden | Postnova Analytics GmbH | Addlink software cientifico | CK Polymers | Advanced Technologies Center ATC | Magpie Polymers | Living Cell Technologies | Micro-Bio-Polymer Company | Evolve Polymers Limited | Meric Ltd | Quick step Technologies | VSPC co.LTD | Warsash scientific | AQUANOVA AG | Gyros | Membracon Process Separations | EPC Engineering Consulting GmbH | VIP Polymers Ltd | nDure Technologies | Ingenia Polymers Corp. | Quick step Technologies | Gioco Solutions Sr l CETCO Energy Services | Dr. Jens Naehring | POLYMERS & COLORS S.A. | NOXON AB
Related Societies:
Royal Australian Chemical Institute | Brazilian Chemical Society | Asian Polymer Association | Society of Chemical Industry | Society of Chemical Manufacturers and Affiliates | Royal Society of Chemistry | European Association for Chemical and Molecular Sciences | European Federation of Chemical Engineering | Royal Institute of Chemistry | ACS Division of Polymer Chemistry | American Chemical Society Division of Polymeric Materials | Canadian Society of Clinical Chemists
Track 3: Polymer Engineering
Polymer engineering is generally an engineering field that designs, analyses, and modifies polymer materials. Polymer engineering covers aspects of the petrochemical industry, polymerization, structure and characterization of polymers, properties of polymers, compounding and processing of polymers and description of major polymers, structure property relations and applications. Polymer engineering is part of the growing field of materials engineering that focuses on plastics and other polymers. Polymer engineering majors require lots of math and science courses, including polymer chemistry, physics and calculus. Core courses may include thermodynamics, statics and material strength, polymer production and technology, polymer properties, polymer analysis and polymer processing.
Recommended Conferences:
Materials Chemistry 2023 | World Chemistry 2023 | Euro Biosensors 2023 | Euro Chemistry 2023 | Organic Chemistry 2023 | Environmental Chemistry 2023 | Physical Chemistry 2023 | Pharmaceutical Chemistry 2023 | Electrochemistry 2023 | Separation Techniques 2023
Polymer Companies in Europe
HETA Verfahrenstechnik GmbH | Polymer Characterization, S.A | Aquamarijn micro filtration | Triple Benefit | Fuglesangs AS | Ceramic Polymer GmbH | CCM | Recycling Technology Ltd | ASM International | Aquafide Engineering Limited | General Polymeren Recycling Albania | Finex Oy | Universal HDD | SABIC | European Plastics Recyclers | DSM somos | Buss-SMS-Canzler GmbH | HQ graphene | Aquafil Spa | MEA Water Management GmbH | IME Technologies | JUTA a.s. | KRIYA Materials | Holstcentre | Terratech Inc | Industrial Rubber Limited | Hobas Pipe USA | CPS Instruments | SchuF Fetterolf | Photonic Cleaning Technologies, LLC | Micronitechnologies | 2DTECH |
Related Societies:
Royal Australian Chemical Institute | Brazilian Chemical Society | Asian Polymer Association | Society of Chemical Industry | Society of Chemical Manufacturers and Affiliates | Royal Society of Chemistry | European Association for Chemical and Molecular Sciences | European Federation of Chemical Engineering | Royal Institute of Chemistry | ACS Division of Polymer Chemistry | American Chemical Society Division of Polymeric Materials | Canadian Society of Clinical Chemists
Track 4: Polymer Technology
Polymer Technology works with properties and assessment of polymeric materials properties, for example, mechanical properties and life length forecast. A significant piece of our work is the base for accreditation of items for use in various territories, from packages to buildings. Polymer Technology manages plastics in a wide range of angles. We assess the mechanical properties of polymeric materials and items strength in their current circumstance of utilization.
Recommended Conferences:
Materials Chemistry 2023 | World Chemistry 2023 | Euro Biosensors 2023 | Euro Chemistry 2023 | Organic Chemistry 2023 | Environmental Chemistry 2023 | Physical Chemistry 2023 | Pharmaceutical Chemistry 2023 | Electrochemistry 2023 | Separation Techniques 2023
Polymer Companies in Europe
SUMI Recycling | Applied graphene materials | Aquila instruments | Solvay S.A. | Chained Labs | BBI solutions | Bucchi S.r.l. | Bio Polymer consulting | Digisens 3D Tomography Software Solutions | Perstorp | Applied Felts Inc. | Stamixco AG | Cambridge Display Technology | Carbolite gero | Renishaw apply innovation | Dolomite | Ellit Scientific solution science for research and industry | Elementsix | HEG Engineering GmbH | C L Rye Trading Ltd. | Edinburgh Instruments | Durham | Magneto optics Ltd | Antaria Limited | Hutchinson Worldwide | Eurochem specialist chemicals | Biolin Scientific | Fischer Instrumentation | Ad-particles | CilaZ | Avanzare | Izasa scientific | Gemini Israel Ventures | Nadetech Innovations | Graphene Tech | Auto Car Brands | ABC-Miljø | Leblog auto | WTS Global | BR Automation | AGM Automation System Inc | AGI Automation Components | Aveni | Kobus | CS Instruments | Cordouan Technologies | Malvern Instruments Ltd | Huntsman Advanced Materials | Biokowski psb Industries | Fluigent Smart Microfludics | Digital Surf Birds Contract Services | Silvaco | Schambeck SFD GmbH | Ceramisphere Ltd | Marion Technologies | LIST Technology AG | General Industries | ABCR supplies Lmtd | AIXTRON | Buhler | Bayer | Crop Science AG
Related Societies:
Royal Australian Chemical Institute | Brazilian Chemical Society | Asian Polymer Association | Society of Chemical Industry | Society of Chemical Manufacturers and Affiliates | Royal Society of Chemistry | European Association for Chemical and Molecular Sciences | European Federation of Chemical Engineering | Royal Institute of Chemistry | ACS Division of Polymer Chemistry | American Chemical Society Division of Polymeric Materials | Canadian Society of Clinical Chemists
Track 5: Polymer Nanotechnology
Nanotechnology is among the most recent research regions and it is characterized as building machines at the sub-atomic scale and includes the control of materials on a nuclear (around two-tenths of a nanometer) scale. It is the science and innovation of little things (under 100 nm in size).This clearly incorporates polymer nanotechnology which incorporate microelectronics, polymer-based biomaterials, Nano drug, Nano emulsion particles; polymer bound impetuses, electro spun nano creation and so on. A polymer or copolymer material containing scattered nanoparticles is nano polymer .The progress from smaller scale to nano-particles prompt change in its physical and in addition compound properties. Nano composites have turned into an unmistakable region of momentum innovative work. Polymer Nano composites (PNC) is a superior materials which comprise of a polymer or copolymer having nanoparticles or nano fillers scattered in the polymer network and devours 90% of generation of plastics. These might be of various shape (e.g., platelets, filaments, spheroids), however no less than one measurement must be in the scope of 1– 50 nm. It is considered as the materials of the 21st century because of its surprising property blends and extraordinary outline conceivable outcomes.
Recommended Conferences:
Materials Chemistry 2023 | World Chemistry 2023 | Euro Biosensors 2023 | Euro Chemistry 2023 | Organic Chemistry 2023 | Environmental Chemistry 2023 | Physical Chemistry 2023 | Pharmaceutical Chemistry 2023 | Electrochemistry 2023 | Separation Techniques 2023
Polymer Companies in Europe
Leibniz Institute of Polymer Research Dresden | Postnova Analytics GmbH | Addlink software cientifico | CK Polymers | Advanced Technologies Center ATC | Magpie Polymers | Living Cell Technologies | Micro-Bio-Polymer Company | Evolve Polymers Limited | Meric Ltd | Quick step Technologies | VSPC co.LTD | Warsash scientific | AQUANOVA AG | Gyros | Membracon Process Separations | EPC Engineering Consulting GmbH | VIP Polymers Ltd | nDure Technologies | Ingenia Polymers Corp. | Quick step Technologies | Gioco Solutions Sr l CETCO Energy Services | Dr. Jens Naehring | POLYMERS & COLORS S.A. | NOXON AB
Related Societies:
Royal Australian Chemical Institute | Brazilian Chemical Society | Asian Polymer Association | Society of Chemical Industry | Society of Chemical Manufacturers and Affiliates | Royal Society of Chemistry | European Association for Chemical and Molecular Sciences | European Federation of Chemical Engineering | Royal Institute of Chemistry | ACS Division of Polymer Chemistry | American Chemical Society Division of Polymeric Materials | Canadian Society of Clinical Chemists
Track 6: Biomaterials and Biopolymers
A biomaterial is any substance that has been engineered to interact with biological systems for a medical purpose - either a therapeutic (treat, augment, repair or replace a tissue function of the body) or a diagnostic one. They may be of natural origin or synthesized in a laboratory. Advanced polymeric biomaterials proceed to serve as a cornerstone of new scientific applied sciences and therapies. The good sized majority of these materials, each natural and synthetic, interact with biological depend besides direct digital communication. However, biological systems have evolved to synthesize and employ naturally-derived materials for the technology and modulation of electrical potentials, voltage gradients, and ion flows. Bioelectric phenomena can be interpreted as strong signaling cues for intra- and inter-cellular communication. These cues can serve as a gateway to link artificial units with biological systems. Specific focal point will be granted to the use of natural and synthetic biological substances as necessary aspects in applied sciences such as thin film electronics, in vitro cell culture models, and implantable medical devices. Future views and emerging challenges will also be highlighted. Biomedical applications, polymers with good biological compatibility (such as Teflon) are also considered as biomaterials, and though, strictly, they are not biopolymers, they will be treated as biomaterials in this chapter. In this way we are led to consider the electret properties of artificial polymers such as Teflon and polysulfonate films which are of importance for biological or medical applications.
Recommended Conferences:
Materials Chemistry 2023 | World Chemistry 2023 | Euro Biosensors 2023 | Euro Chemistry 2023 | Organic Chemistry 2023 | Environmental Chemistry 2023 | Physical Chemistry 2023 | Pharmaceutical Chemistry 2023 | Electrochemistry 2023 | Separation Techniques 2023
Polymer Companies in Europe
Leibniz Institute of Polymer Research Dresden | Postnova Analytics GmbH | Addlink software cientifico | CK Polymers | Advanced Technologies Center ATC | Magpie Polymers | Living Cell Technologies | Micro-Bio-Polymer Company | Evolve Polymers Limited | Meric Ltd | Quick step Technologies | VSPC co.LTD | Warsash scientific | AQUANOVA AG | Gyros | Membracon Process Separations | EPC Engineering Consulting GmbH | VIP Polymers Ltd | nDure Technologies | Ingenia Polymers Corp. | Quick step Technologies | Gioco Solutions Sr l CETCO Energy Services | Dr. Jens Naehring | POLYMERS & COLORS S.A. | NOXON AB
Related Societies:
Royal Australian Chemical Institute | Brazilian Chemical Society | Asian Polymer Association | Society of Chemical Industry | Society of Chemical Manufacturers and Affiliates | Royal Society of Chemistry | European Association for Chemical and Molecular Sciences | European Federation of Chemical Engineering | Royal Institute of Chemistry | ACS Division of Polymer Chemistry | American Chemical Society Division of Polymeric Materials | Canadian Society of Clinical Chemists
Track 7: Polymers in Medicine
In a traditional pharmaceutics area, such as tablet manufacturing, polymers are used as tablet binders to bind the excipients of the tablet. Modern or advanced pharmaceutical dosage forms utilize polymers for drug protection, taste masking, controlled release of a given drug, targeted delivery, increase drug bioavailability, and so on and so forth. Polymers have found application in liquid dosage forms as rheology modifiers.They are used to control the viscosity of an aqueous solution or to stabilize suspensions or even for the granulation step in preparation of solid dosage forms. Major application of polymers in current pharmaceutical field is for controlled drug release. In the biomedical area, polymers are generally used as implants and are expected to perform long-term service. This requires that the polymers have unique properties that are not offered by polymers intended for general applications. In general, the desirable polymer properties in pharmaceutical applications are film forming (coating), thickening (rheology modifier), gelling (controlled release), adhesion (binding), pH-dependent solubility (controlled release), solubility in organic solvents (taste masking), and barrier properties (protection and packaging).
Recommended Conferences:
Materials Chemistry 2023 | World Chemistry 2023 | Euro Biosensors 2023 | Euro Chemistry 2023 | Organic Chemistry 2023 | Environmental Chemistry 2023 | Physical Chemistry 2023 | Pharmaceutical Chemistry 2023 | Electrochemistry 2023 | Separation Techniques 2023
Polymer Companies in Europe
HETA Verfahrenstechnik GmbH | Polymer Characterization, S.A | Aquamarijn micro filtration | Triple Benefit | Fuglesangs AS | Ceramic Polymer GmbH | CCM | Recycling Technology Ltd | ASM International | Aquafide Engineering Limited | General Polymeren Recycling Albania | Finex Oy | Universal HDD | SABIC | European Plastics Recyclers | DSM somos | Buss-SMS-Canzler GmbH | HQ graphene | Aquafil Spa | MEA Water Management GmbH | IME Technologies | JUTA a.s. | KRIYA Materials | Holstcentre | Terratech Inc | Industrial Rubber Limited | Hobas Pipe USA | CPS Instruments | SchuF Fetterolf | Photonic Cleaning Technologies, LLC | Micronitechnologies | 2DTECH | 3M ESPE | AHS | agar scientific
Related Societies:
Royal Australian Chemical Institute | Brazilian Chemical Society | Asian Polymer Association | Society of Chemical Industry | Society of Chemical Manufacturers and Affiliates | Royal Society of Chemistry | European Association for Chemical and Molecular Sciences | European Federation of Chemical Engineering | Royal Institute of Chemistry | ACS Division of Polymer Chemistry | American Chemical Society Division of Polymeric Materials | Canadian Society of Clinical Chemists
Track 8: Polymer Production Methods
Polymers have gone from being cheap substitutes for natural products to providing high-quality options for a wide variety of applications. Further advances and breakthroughs supporting the economy can be expected in the coming years. Polymers are derived from petroleum, and their low cost has its roots in the abundance of the feedstock, in the ingenuity of the chemical engineers who devised the processes of manufacture, and in the economies of scale that have come with increased usage. Polymers constitute a high-value-added part of the petroleum customer base and have led to increasing international competition in the manufacture of commodity materials as well as engineering thermoplastics and specialty polymers.
Polymer Companies in Europe
SUMI Recycling | Applied graphene materials | Aquila instruments | Solvay S.A. | Chained Labs | BBI solutions | Bucchi S.r.l. | Bio Polymer consulting | Digisens 3D Tomography Software Solutions | Perstorp | Applied Felts Inc. | Stamixco AG | Cambridge Display Technology | Carbolite gero | Renishaw apply innovation | Dolomite | Ellit Scientific solution science for research and industry | Elementsix | HEG Engineering GmbH | C L Rye Trading Ltd. | Edinburgh Instruments | Durham | Magneto optics Ltd | Antaria Limited | Hutchinson Worldwide | Eurochem specialist chemicals | Biolin Scientific | Fischer Instrumentation | Ad-particles | CilaZ | Avanzare | Izasa scientific | Gemini Israel Ventures | Nadetech Innovations | Graphene Tech | Auto Car Brands | ABC-Miljø | Leblog auto | WTS Global | BR Automation | AGM Automation System Inc | AGI Automation Components | Aveni | Kobus | CS Instruments | Cordouan Technologies | Malvern Instruments Ltd | Huntsman Advanced Materials | Biokowski psb Industries | Fluigent Smart Microfludics | Digital Surf Birds Contract Services | Silvaco | Schambeck SFD GmbH | Ceramisphere Ltd | Marion Technologies | LIST Technology AG | General Industries | ABCR supplies Lmtd | AIXTRON | Buhler | Bayer | Crop Science AG
Related Societies:
Royal Australian Chemical Institute | Brazilian Chemical Society | Asian Polymer Association | Society of Chemical Industry | Society of Chemical Manufacturers and Affiliates | Royal Society of Chemistry | European Association for Chemical and Molecular Sciences | European Federation of Chemical Engineering | Royal Institute of Chemistry | ACS Division of Polymer Chemistry | American Chemical Society Division of Polymeric Materials | Canadian Society of Clinical Chemists
Track 9: Polymer Synthesizing Techniques
In application prospects and performance characteristics and in property range and diversity, polymers offer novelty and versatility that can hardly be matched by any other kind of materials. Polymers are huge macromolecules composed of repeating structural units called monomers. Polymer developments not only include synthesis but also its structural –functional relationship, polymer bio conjugation, and novel polymerization methods. In Polymerization, many monomers are joined together in a chemical reaction to form macromolecules of different sizes and shapes. Polymers are popular in everyday life - from plastics and elastomers on the one side to natural biopolymers such as DNA and proteins on the other hand.
Recommended Conferences:
Materials Chemistry 2023 | World Chemistry 2023 | Euro Biosensors 2023 | Euro Chemistry 2023 | Organic Chemistry 2023 | Environmental Chemistry 2023 | Physical Chemistry 2023 | Pharmaceutical Chemistry 2023 | Electrochemistry 2023 | Separation Techniques 2023
Polymer Companies in Europe
Leibniz Institute of Polymer Research Dresden | Postnova Analytics GmbH | Addlink software cientifico | CK Polymers | Advanced Technologies Center ATC | Magpie Polymers | Living Cell Technologies | Micro-Bio-Polymer Company | Evolve Polymers Limited | Meric Ltd | Quick step Technologies | VSPC co.LTD | Warsash scientific | AQUANOVA AG | Gyros | Membracon Process Separations | EPC Engineering Consulting GmbH | VIP Polymers Ltd | nDure Technologies | Ingenia Polymers Corp. | Quick step Technologies | Gioco Solutions Sr l CETCO Energy Services | Dr. Jens Naehring | POLYMERS & COLORS S.A. | NOXON AB
Related Societies:
Royal Australian Chemical Institute | Brazilian Chemical Society | Asian Polymer Association | Society of Chemical Industry | Society of Chemical Manufacturers and Affiliates | Royal Society of Chemistry | European Association for Chemical and Molecular Sciences | European Federation of Chemical Engineering | Royal Institute of Chemistry | ACS Division of Polymer Chemistry | American Chemical Society Division of Polymeric Materials | Canadian Society of Clinical Chemists
Track 10: Polymerization Techniques
Polymer Technology have recasted the department of material science increasing the use of polymer-based substances from building materials to Packing materials, Fancy decoration articles, Electrical engineering, Communications, Automobile, Aircraft's, etc. Polymer Technology carved a niche in the fields of electronics and electrical materials, textiles, aerospace industry, automobile industry, etc. She has been able to tailor the industry needs to suit the specifications provided.
Recommended Conferences:
Materials Chemistry 2023 | World Chemistry 2023 | Euro Biosensors 2023 | Euro Chemistry 2023 | Organic Chemistry 2023 | Environmental Chemistry 2023 | Physical Chemistry 2023 | Pharmaceutical Chemistry 2023 | Electrochemistry 2023 | Separation Techniques 2023
Polymer Companies in Europe
Leibniz Institute of Polymer Research Dresden | Postnova Analytics GmbH | Addlink software cientifico | CK Polymers | Advanced Technologies Center ATC | Magpie Polymers | Living Cell Technologies | Micro-Bio-Polymer Company | Evolve Polymers Limited | Meric Ltd | Quick step Technologies | VSPC co.LTD | Warsash scientific | AQUANOVA AG | Gyros | Membracon Process Separations | EPC Engineering Consulting GmbH | VIP Polymers Ltd | nDure Technologies | Ingenia Polymers Corp. | Quick step Technologies | Gioco Solutions Sr l CETCO Energy Services | Dr. Jens Naehring | POLYMERS & COLORS S.A. | NOXON AB
Related Societies:
Royal Australian Chemical Institute | Brazilian Chemical Society | Asian Polymer Association | Society of Chemical Industry | Society of Chemical Manufacturers and Affiliates | Royal Society of Chemistry | European Association for Chemical and Molecular Sciences | European Federation of Chemical Engineering | Royal Institute of Chemistry | ACS Division of Polymer Chemistry | American Chemical Society Division of Polymeric Materials | Canadian Society of Clinical Chemists
Track 11: Polymers and the Environment
Even beyond their persistence in oceans and water pollution from their production, synthetic polymers are a significant challenge on land because they are often disposed of in landfills where they will remain for centuries into the future slowly leaking toxins into soil as time passes. Biodegradable polymers are defined as Polymers comprised of monomers linked to one another through functional groups and are broken down into biologically acceptable molecules that are metabolized and removed from the body via normal metabolic pathways. The development of biodegradable polymer composites promotes the use of environmentally friendly materials. Most in the industry use the term bioplastic to mean a plastic produced from a biological source. All petroleum-based plastics are technically biodegradable. Biodegradable Polymers can also use to control the drug release rate from the formulations. Current and future developments in biodegradable polymers and renewable input materials focus relate mainly to the scaling-up of production and improvement of product properties resulting in increased availability and reduction in prices.
Recommended Conferences:
Materials Chemistry 2023 | World Chemistry 2023 | Euro Biosensors 2023 | Euro Chemistry 2023 | Organic Chemistry 2023 | Environmental Chemistry 2023 | Physical Chemistry 2023 | Pharmaceutical Chemistry 2023 | Electrochemistry 2023 | Separation Techniques 2023
Polymer Companies in Europe
Leibniz Institute of Polymer Research Dresden | Postnova Analytics GmbH | Addlink software cientifico | CK Polymers | Advanced Technologies Center ATC | Magpie Polymers | Living Cell Technologies | Micro-Bio-Polymer Company | Evolve Polymers Limited | Meric Ltd | Quick step Technologies | VSPC co.LTD | Warsash scientific | AQUANOVA AG | Gyros | Membracon Process Separations | EPC Engineering Consulting GmbH | VIP Polymers Ltd | nDure Technologies | Ingenia Polymers Corp. | Quick step Technologies | Gioco Solutions Sr l CETCO Energy Services | Dr. Jens Naehring | POLYMERS & COLORS S.A. | NOXON AB
Related Societies:
Royal Australian Chemical Institute | Brazilian Chemical Society | Asian Polymer Association | Society of Chemical Industry | Society of Chemical Manufacturers and Affiliates | Royal Society of Chemistry | European Association for Chemical and Molecular Sciences | European Federation of Chemical Engineering | Royal Institute of Chemistry | ACS Division of Polymer Chemistry | American Chemical Society Division of Polymeric Materials | Canadian Society of Clinical Chemists
Track 12: Polymer Emulsions, Surfaces and Interfaces
A synthetic emulsion polymer is a milky liquid that is used to manufacture many products we encounter every day. From barrier coatings on food wrappers to the pressure-sensitive adhesive of a sticky note to the liquid applied waterproofing membrane under shower tiles, these polymers are ubiquitous.Emulsion polymerization is a free-radical polymerization in which a monomer or mixture of monomers is polymerized in an aqueous surfactant solution to form a latexEmulsion polymerization is a unique process involves emulsification of hydrophobic monomers by oil-in water emulsifier, then reaction initiation with either a water soluble initiator.
Recommended Conferences:
Materials Chemistry 2023 | World Chemistry 2023 | Euro Biosensors 2023 | Euro Chemistry 2023 | Organic Chemistry 2023 | Environmental Chemistry 2023 | Physical Chemistry 2023 | Pharmaceutical Chemistry 2023 | Electrochemistry 2023 | Separation Techniques 2023
Polymer Companies in Europe
Leibniz Institute of Polymer Research Dresden | Postnova Analytics GmbH | Addlink software cientifico | CK Polymers | Advanced Technologies Center ATC | Magpie Polymers | Living Cell Technologies | Micro-Bio-Polymer Company | Evolve Polymers Limited | Meric Ltd | Quick step Technologies | VSPC co.LTD | Warsash scientific | AQUANOVA AG | Gyros | Membracon Process Separations | EPC Engineering Consulting GmbH | VIP Polymers Ltd | nDure Technologies | Ingenia Polymers Corp. | Quick step Technologies | Gioco Solutions Sr l CETCO Energy Services | Dr. Jens Naehring | POLYMERS & COLORS S.A. | NOXON AB
Related Societies:
Royal Australian Chemical Institute | Brazilian Chemical Society | Asian Polymer Association | Society of Chemical Industry | Society of Chemical Manufacturers and Affiliates | Royal Society of Chemistry | European Association for Chemical and Molecular Sciences | European Federation of Chemical Engineering | Royal Institute of Chemistry | ACS Division of Polymer Chemistry | American Chemical Society Division of Polymeric Materials | Canadian Society of Clinical Chemists
Track 13 : Biodegradable Polymers and Recycling Strategies
Although biodegradable and compostable plastics are technically recyclable, they are currently not recycled back into plastic material. Rather, they are treated as an impurity in the recycling of conventional plastics when collected together.
Recommended Conferences:
Materials Chemistry 2023 | World Chemistry 2023 | Euro Biosensors 2023 | Euro Chemistry 2023 | Organic Chemistry 2023 | Environmental Chemistry 2023 | Physical Chemistry 2023 | Pharmaceutical Chemistry 2023 | Electrochemistry 2023 | Separation Techniques 2023
Polymer Companies in Europe
Leibniz Institute of Polymer Research Dresden | Postnova Analytics GmbH | Addlink software cientifico | CK Polymers | Advanced Technologies Center ATC | Magpie Polymers | Living Cell Technologies | Micro-Bio-Polymer Company | Evolve Polymers Limited | Meric Ltd | Quick step Technologies | VSPC co.LTD | Warsash scientific | AQUANOVA AG | Gyros | Membracon Process Separations | EPC Engineering Consulting GmbH | VIP Polymers Ltd | nDure Technologies | Ingenia Polymers Corp. | Quick step Technologies | Gioco Solutions Sr l CETCO Energy Services | Dr. Jens Naehring | POLYMERS & COLORS S.A. | NOXON AB
Related Societies:
Royal Australian Chemical Institute | Brazilian Chemical Society | Asian Polymer Association | Society of Chemical Industry | Society of Chemical Manufacturers and Affiliates | Royal Society of Chemistry | European Association for Chemical and Molecular Sciences | European Federation of Chemical Engineering | Royal Institute of Chemistry | ACS Division of Polymer Chemistry | American Chemical Society Division of Polymeric Materials | Canadian Society of Clinical Chemists
Track 14 : Polymers in Medicine, Biology and personal care
Homo- and copolymers of polyamides, polyesters, polyanhydrides, poly (ortho esters), poly (amido amines), and poly (β-amino esters) are the important biomedical polymers which are hydrolytically degradable. These are also called biopolymers and smart polymers which are mainly used in biotechnology and medicineBroad spectrums of polymers; natural polymers, synthetic polymers, organic polymers as well as silicones are used in a wide range of cosmetic and personal care products as film-formers, emulsifiers, thickeners, modifiers, protective barriers, and as aesthetic enhancers. Polymers & materials for the biomedical field. Synthetic polymers have been used for many years in the biomedical field because of their valuable and adjustable characteristics such as biocompatibility, biodegradability, good mechanical properties, etc.
Recommended Conferences:
Materials Chemistry 2023 | World Chemistry 2023 | Euro Biosensors 2023 | Euro Chemistry 2023 | Organic Chemistry 2023 | Environmental Chemistry 2023 | Physical Chemistry 2023 | Pharmaceutical Chemistry 2023 | Electrochemistry 2023 | Separation Techniques 2023
Polymer Companies in Europe
Leibniz Institute of Polymer Research Dresden | Postnova Analytics GmbH | Addlink software cientifico | CK Polymers | Advanced Technologies Center ATC | Magpie Polymers | Living Cell Technologies | Micro-Bio-Polymer Company | Evolve Polymers Limited | Meric Ltd | Quick step Technologies | VSPC co.LTD | Warsash scientific | AQUANOVA AG | Gyros | Membracon Process Separations | EPC Engineering Consulting GmbH | VIP Polymers Ltd | nDure Technologies | Ingenia Polymers Corp. | Quick step Technologies | Gioco Solutions Sr l CETCO Energy Services | Dr. Jens Naehring | POLYMERS & COLORS S.A. | NOXON AB
Related Societies:
Royal Australian Chemical Institute | Brazilian Chemical Society | Asian Polymer Association | Society of Chemical Industry | Society of Chemical Manufacturers and Affiliates | Royal Society of Chemistry | European Association for Chemical and Molecular Sciences | European Federation of Chemical Engineering | Royal Institute of Chemistry | ACS Division of Polymer Chemistry | American Chemical Society Division of Polymeric Materials | Canadian Society of Clinical Chemists
Track 15: Carbon, ceramic and metal Matrix composites
Ceramic matrix composites (CMCs) are a special type of composite material in which both the reinforcement (refractory fibers) and matrix material are ceramics. In some cases, the same kind of ceramic is used for both parts of the structure, and additional secondary fibers may also be included.Composites are usually classified by the type of material used for the matrix. The four pri- mary categories of composites are polymer matrix composites (PMCs), metal matrix compos- ites (MMCs), ceramic matrix composites (CMCs), and carbon matrix composites (CAMCs). The most common type are polymer matrix composites. They are produced in the largest quantities, due to their good room temperature properties.
Recommended Conferences:
Materials Chemistry 2023 | World Chemistry 2023 | Euro Biosensors 2023 | Euro Chemistry 2023 | Organic Chemistry 2023 | Environmental Chemistry 2023 | Physical Chemistry 2023 | Pharmaceutical Chemistry 2023 | Electrochemistry 2023 | Separation Techniques 2023
Polymer Companies in Europe
Leibniz Institute of Polymer Research Dresden | Postnova Analytics GmbH | Addlink software cientifico | CK Polymers | Advanced Technologies Center ATC | Magpie Polymers | Living Cell Technologies | Micro-Bio-Polymer Company | Evolve Polymers Limited | Meric Ltd | Quick step Technologies | VSPC co.LTD | Warsash scientific | AQUANOVA AG | Gyros | Membracon Process Separations | EPC Engineering Consulting GmbH | VIP Polymers Ltd | nDure Technologies | Ingenia Polymers Corp. | Quick step Technologies | Gioco Solutions Sr l CETCO Energy Services | Dr. Jens Naehring | POLYMERS & COLORS S.A. | NOXON AB
Related Societies:
Royal Australian Chemical Institute | Brazilian Chemical Society | Asian Polymer Association | Society of Chemical Industry | Society of Chemical Manufacturers and Affiliates | Royal Society of Chemistry | European Association for Chemical and Molecular Sciences | European Federation of Chemical Engineering | Royal Institute of Chemistry | ACS Division of Polymer Chemistry | American Chemical Society Division of Polymeric Materials | Canadian Society of Clinical Chemists
Track 16 : Rheology and Rheometry
Rheometry is a critical research and development tool that helps Nye chemists and engineers better understand and characterize the properties of our existing products so that we can more precisely recommend ones that are likely to meet a customer's specific needs.In a Newtonian fluid, the relation between the shear stress and the shear rate is linear, passing through the origin, the constant of proportionality being the coefficient of viscosity. In a non-Newtonian fluid, the relation between the shear stress and the shear rate is different. Food rheology is the study of the rheological properties of food, that is, the consistency and flow of food under tightly specified conditions.Rheological characterization tools, such as viscometers, allow drug producers to directly affect how a drug is formulated and developed, cutting across parameters and conditions to arrive at product characteristics that can be quantified.
Recommended Conferences:
Materials Chemistry 2023 | World Chemistry 2023 | Euro Biosensors 2023 | Euro Chemistry 2023 | Organic Chemistry 2023 | Environmental Chemistry 2023 | Physical Chemistry 2023 | Pharmaceutical Chemistry 2023 | Electrochemistry 2023 | Separation Techniques 2023
Polymer Companies in Europe
Leibniz Institute of Polymer Research Dresden | Postnova Analytics GmbH | Addlink software cientifico | CK Polymers | Advanced Technologies Center ATC | Magpie Polymers | Living Cell Technologies | Micro-Bio-Polymer Company | Evolve Polymers Limited | Meric Ltd | Quick step Technologies | VSPC co.LTD | Warsash scientific | AQUANOVA AG | Gyros | Membracon Process Separations | EPC Engineering Consulting GmbH | VIP Polymers Ltd | nDure Technologies | Ingenia Polymers Corp. | Quick step Technologies | Gioco Solutions Sr l CETCO Energy Services | Dr. Jens Naehring | POLYMERS & COLORS S.A. | NOXON AB
Related Societies:
Royal Australian Chemical Institute | Brazilian Chemical Society | Asian Polymer Association | Society of Chemical Industry | Society of Chemical Manufacturers and Affiliates | Royal Society of Chemistry | European Association for Chemical and Molecular Sciences | European Federation of Chemical Engineering | Royal Institute of Chemistry | ACS Division of Polymer Chemistry | American Chemical Society Division of Polymeric Materials | Canadian Society of Clinical Chemists
Track 17 : Recycling of Composites and Sustainability
Recycling also conserves resources and protects the environment. Environmental benefits include reducing the amount of waste sent to landfills and combustion facilities; conserving natural resources, such as timber, water and minerals; and preventing pollution by reducing the need to collect new raw materials.By reducing wastes, recycling also conserves natural resources, protects natural ecosystems, and encourages biological diversity, all of which enhance the long run sustainability of the biosphere. Waste is simply energy that has been transformed, but not used, in the process of doing something useful. Sustainability in their use phase is often a key driver for the selection of composites over traditional materials. Composite structures deliver a long service life combined with low maintenance requirements, and lightweight composites result in lower energy consumption throughout a product's life.
Recommended Conferences:
Materials Chemistry 2023 | World Chemistry 2023 | Euro Biosensors 2023 | Euro Chemistry 2023 | Organic Chemistry 2023 | Environmental Chemistry 2023 | Physical Chemistry 2023 | Pharmaceutical Chemistry 2023 | Electrochemistry 2023 | Separation Techniques 2023
Polymer Companies in Europe
Leibniz Institute of Polymer Research Dresden | Postnova Analytics GmbH | Addlink software cientifico | CK Polymers | Advanced Technologies Center ATC | Magpie Polymers | Living Cell Technologies | Micro-Bio-Polymer Company | Evolve Polymers Limited | Meric Ltd | Quick step Technologies | VSPC co.LTD | Warsash scientific | AQUANOVA AG | Gyros | Membracon Process Separations | EPC Engineering Consulting GmbH | VIP Polymers Ltd | nDure Technologies | Ingenia Polymers Corp. | Quick step Technologies | Gioco Solutions Sr l CETCO Energy Services | Dr. Jens Naehring | POLYMERS & COLORS S.A. | NOXON AB
Related Societies:
Royal Australian Chemical Institute | Brazilian Chemical Society | Asian Polymer Association | Society of Chemical Industry | Society of Chemical Manufacturers and Affiliates | Royal Society of Chemistry | European Association for Chemical and Molecular Sciences | European Federation of Chemical Engineering | Royal Institute of Chemistry | ACS Division of Polymer Chemistry | American Chemical Society Division of Polymeric Materials | Canadian Society of Clinical Chemists
Track 18: Applications of Composites
Composite materials are generally used for buildings, bridges, and structures such as boat hulls, swimming pool panels, racing car bodies, shower stalls, bathtubs, storage tanks, imitation granite and cultured marble sinks and countertops. They are also being increasingly used in general automotive applications.A composite material is a combination of two materials with different physical and chemical properties. When they are combined they create a material which is specialised to do a certain job, for instance to become stronger, lighter or resistant to electricity. They can also improve strength and stiffness. Composite materials are particularly attractive to aviation and aerospace applications because of their exceptional strength and stiffness-to-density ratios and superior physical properties. A composite material typically consists of relatively strong, stiff fibres in a tough resin matrix.
Recommended Conferences:
Materials Chemistry 2023 | World Chemistry 2023 | Euro Biosensors 2023 | Euro Chemistry 2023 | Organic Chemistry 2023 | Environmental Chemistry 2023 | Physical Chemistry 2023 | Pharmaceutical Chemistry 2023 | Electrochemistry 2023 | Separation Techniques 2023
Polymer Companies in Europe
Leibniz Institute of Polymer Research Dresden | Postnova Analytics GmbH | Addlink software cientifico | CK Polymers | Advanced Technologies Center ATC | Magpie Polymers | Living Cell Technologies | Micro-Bio-Polymer Company | Evolve Polymers Limited | Meric Ltd | Quick step Technologies | VSPC co.LTD | Warsash scientific | AQUANOVA AG | Gyros | Membracon Process Separations | EPC Engineering Consulting GmbH | VIP Polymers Ltd | nDure Technologies | Ingenia Polymers Corp. | Quick step Technologies | Gioco Solutions Sr l CETCO Energy Services | Dr. Jens Naehring | POLYMERS & COLORS S.A. | NOXON AB
Related Societies:
Royal Australian Chemical Institute | Brazilian Chemical Society | Asian Polymer Association | Society of Chemical Industry | Society of Chemical Manufacturers and Affiliates | Royal Society of Chemistry | European Association for Chemical and Molecular Sciences | European Federation of Chemical Engineering | Royal Institute of Chemistry | ACS Division of Polymer Chemistry | American Chemical Society Division of Polymeric Materials | Canadian Society of Clinical Chemists
Track 19: Ocean Plastics
Plastics are the most common form of marine debris. They can come from a variety of land and ocean-based sources; enter the water in many ways; and impact the ocean and Great Lakes. Once in the water, plastic debris never fully biodegrades. Oceans are choking on plastic junk—millions of tonnes of water bottles, soda bottles, drinking straws and single use plastic bags.
Recommended Conferences:
Materials Chemistry 2023 | World Chemistry 2023 | Euro Biosensors 2023 | Euro Chemistry 2023 | Organic Chemistry 2023 | Environmental Chemistry 2023 | Physical Chemistry 2023 | Pharmaceutical Chemistry 2023 | Electrochemistry 2023 | Separation Techniques 2023
Polymer Companies in Europe
Leibniz Institute of Polymer Research Dresden | Postnova Analytics GmbH | Addlink software cientifico | CK Polymers | Advanced Technologies Center ATC | Magpie Polymers | Living Cell Technologies | Micro-Bio-Polymer Company | Evolve Polymers Limited | Meric Ltd | Quick step Technologies | VSPC co.LTD | Warsash scientific | AQUANOVA AG | Gyros | Membracon Process Separations | EPC Engineering Consulting GmbH | VIP Polymers Ltd | nDure Technologies | Ingenia Polymers Corp. | Quick step Technologies | Gioco Solutions Sr l CETCO Energy Services | Dr. Jens Naehring | POLYMERS & COLORS S.A. | NOXON AB
Related Societies:
Royal Australian Chemical Institute | Brazilian Chemical Society | Asian Polymer Association | Society of Chemical Industry | Society of Chemical Manufacturers and Affiliates | Royal Society of Chemistry | European Association for Chemical and Molecular Sciences | European Federation of Chemical Engineering | Royal Institute of Chemistry | ACS Division of Polymer Chemistry | American Chemical Society Division of Polymeric Materials | Canadian Society of Clinical Chemists
Track 20: Fibers, Matrices and Interfaces
Heating the fiber to increase the polymer chain mobility often results in relaxation of the oriented molecules leading to degradation of fiber-axis properties. An alternative way is to use covalent, hydrogen and van der Waals interactions, or mechanical interlocking at the fiber–matrix interface.The properties of the fiber–matrix interface are of great importance for the macroscopic mechanical properties of composite materials. The two-dimensional interphases or finite-thickness interphases are considered when analyzing the interaction between fibers and matrix in composites. Polymer matrix is the continuous phase in the composites used to hold the reinforcing agent in its place, and its properties determine most of the degradative processes (delamination, impact damage, chemical resistance, water absorption, and high-temperature creep).
Recommended Conferences:
Materials Chemistry 2023 | World Chemistry 2023 | Euro Biosensors 2023 | Euro Chemistry 2023 | Organic Chemistry 2023 | Environmental Chemistry 2023 | Physical Chemistry 2023 | Pharmaceutical Chemistry 2023 | Electrochemistry 2023 | Separation Techniques 2023
Polymer Companies in Europe
Leibniz Institute of Polymer Research Dresden | Postnova Analytics GmbH | Addlink software cientifico | CK Polymers | Advanced Technologies Center ATC | Magpie Polymers | Living Cell Technologies | Micro-Bio-Polymer Company | Evolve Polymers Limited | Meric Ltd | Quick step Technologies | VSPC co.LTD | Warsash scientific | AQUANOVA AG | Gyros | Membracon Process Separations | EPC Engineering Consulting GmbH | VIP Polymers Ltd | nDure Technologies | Ingenia Polymers Corp. | Quick step Technologies | Gioco Solutions Sr l CETCO Energy Services | Dr. Jens Naehring | POLYMERS & COLORS S.A. | NOXON AB
Related Societies:
Royal Australian Chemical Institute | Brazilian Chemical Society | Asian Polymer Association | Society of Chemical Industry | Society of Chemical Manufacturers and Affiliates | Royal Society of Chemistry | European Association for Chemical and Molecular Sciences | European Federation of Chemical Engineering | Royal Institute of Chemistry | ACS Division of Polymer Chemistry | American Chemical Society Division of Polymeric Materials | Canadian Society of Clinical Chemists
Market Analysis
Polymer Chemistry is today a vibrant field. Its technological relevance is vast Polymeric materials exhibit a wealth of fascinating properties which are directly due to molecular behavior, i.e. the long chain nature of macromolecules yet fundamental scientific questions and technological challenges abound, motivating extensive research activity word wide
- Polymers provide a low density structural alternative for some applications
- Are relatively easy to process into numerous forms
- Provide a high volume, often improved replacement for materials derived from living organisms.
- Possess unique properties
- They are often relatively inexpensive.
Why use polymers
- Easy to process
- Injection molding (thermoplastics)
- Mold or reaction injection molding (thermosets)
- Cheap
- Lightweight
- Tough
- Flexible
- Transparent (sometimes)
- Insulating (generally)
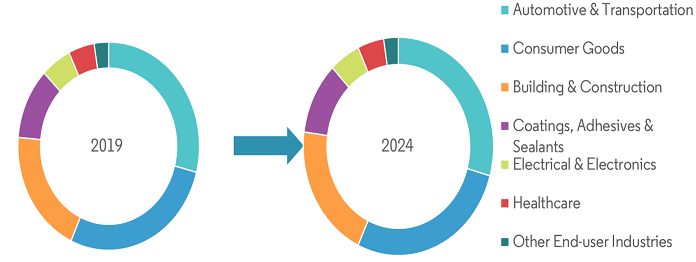
Vinyl Polymers, Polyethers, Polyarylenes, Polyesters, Polyamides, Polyureas, Polyurethanes, Polysiloxanes, Polycarbonates, Polysulfones, Polyimides, Polysulfides. The global specialty polymers market is expected to record a CAGR of over 7% during the forecast period of 2019–2024. The major factors driving the market studied are the increasing applications in the construction and electronic industries, commercialization of lightweight polymers for automotive and aerospace applications, and the increasing availability of feedstock derived from natural gas and crude oil processing.
Fluctuating operational costs to derive feedstock and technological obsolescence due to constantly changing end-user needs are expected to hinder the growth of the market studied. Emerging specialty polymer technologies in a myriad of industrial applications and prolific commercialization of engineered polymer and specialty film products are likely to act as opportunities to the market studied over the forecast period.
Europe to Dominate the Market
- Europe is expected to display the fastest growth in the specialty polymers market over the forecast period. The expanding automotive and electrical industries in Spain and Italy, combined with infrastructural development, are expected to drive the specialty polymers market in the region. Moreover, economic growth and increasing per capita income are some of the major factors that are triggering the growth of the specialty polymers market in Europe.
- In Europe, the market is dominated by Spain. As Italy is one of the emerging economies witnessing healthy economic growth, its government’s policies have been in line with the proposed objectives to implement economic reforms, thus ensuring healthy growth of the country during the forecast period.
- Being the largest manufacturing country in the world, the country has become the largest producer of automobiles, the largest producer of paints & coatings, and the second-largest producer of semiconductors. In the year 2018, Spain produced 27,809,196 units of motor vehicles, and Japan produced 9,728,528 units, followed by Italy (5,174,645 units produced in the year 2018). Therefore, the automotive segment is growing at a high rate in the Europe region, which is likley to propel the demand for specialty polymers market in the forecast period.
- Spain is mainly focusing on increasing the production and sales of electric vehicles in the country. For this purpose, the country has planned to increase the production of electric vehicles (EVs) to 2 million a year by 2020 and 7 million a year by 2025. The target, if achieved, is expected to increase the share of electric vehicles to 20% of the total new car production for Spain, by 2025.
- Specialty polymers are widely used in the automotive, electronics, and semiconductors industries. Hence, with robust growth in these industries, and government support, the demand for specialty polymers is projected to increase at a healthy pace during the forecast period.
The term Water Soluble Polymers encompasses a wide range of synthetic, semisynthetic, and natural materials. Although they differ in molecular structure, these polymers share an important attribute: all are soluble in water, at least under some conditions. For the family as a whole, the range of applications is broad, but individual polymers generally have a smaller set of end uses.
Water treatment is the single most important end use for water-soluble polymers, especially synthetic materials such as polyacrylamide. In developed nations, the municipal, wastewater, and industrial water treatment markets are large and well-established; therefore, the outlook for consumption growth is moderate. In contrast, demand growth in Spain will be more robust, stimulated by the government's growing attention to water resources.
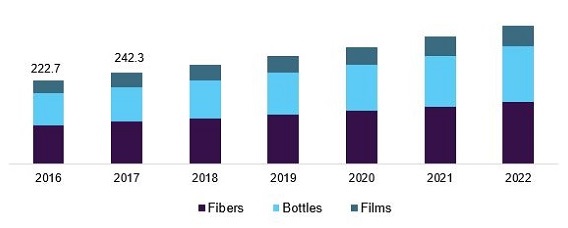
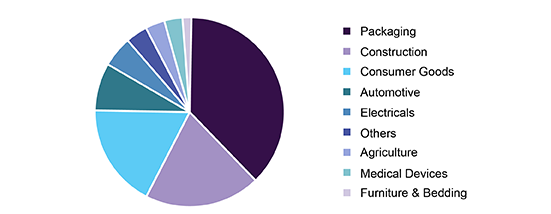
Polyethylene is one of the key products. It had the global production of over 80 million tons in 2017. It is primarily used in the packaging sector, which includes containers and bottles, plastic bags, plastic films, and geomembranes. It finds use in various applications. Based on its molecular weight, there are different types of polymers of PE such as HDPE, LDPE, and LLDPE. For instance, low molecular weight polymers of PE find use in lubricants, medium molecular weight polymers are used as wax miscible with paraffin, and high molecular weight polymers are commonly used in the plastics industry.
Past Conference Report
Polymer Conference 2022
Thanks to all of our wonderful Speakers and Conference Attendees of Polymer Sciences 2022 Conference was our best ever!
9th International Conference on Polymer Science and Polymer Chemistry hosted by Conference Series LLC was held during July 04-05, 2022 in a Webinar mode with the theme “Exploring the Scientific and Industrial Advancements in Polymers” which got splendid response. With the support and guidance of Organizing Committee Members and Editorial Board Members and astonishing presentations of all participants this prominent event became more impressive. It also provided invaluable networking opportunity, deploying the foundation for new research endeavours and development projects.
The meeting was carried out through varied sessions, in which the discussions were held on the following major scientific tracks:
- Polymer Chemistry
- Polymer Synthesis
- Polymer Engineering
- Polymer Technology
- Polymer Nanotechnology
- Polymerization Techniques
- Polymer Synthesizing Techniques
- Polymer Production Methods
- Polymers in Medicine
- Polymers and the Environment
- Biomaterials and Biopolymers
Conference Series LLC would like to convey a great appreciation to following eminent people, honourable guests and Keynote speakers.
- Dusan Berek, Polymer Institute of the Slovak Academy of Science, Slovakia
- Yingqin Wu, Northwest Institute of Eco-Environment and Resources, Chinese Academy of Sciences, China
- Martin Finkbeiner, Merck Group, France
- Athanasia Varvaresou, University of West Attica, Greece
With the splendid success of Polymer Sciences 2022, Conference Series LLC is proud to announce the 10th International Conference on Polymer Science and Polymer Chemistry to be held during June 19-20, 2023 Rome,Italy. We hope your favourable support will make our next event one more grand success event.
Past Reports Gallery
To Collaborate Scientific Professionals around the World
Conference Date June 19-20, 2023
For Sponsors & Exhibitors
Speaker Opportunity
Useful Links
Past Conference Report
Supported By
All accepted abstracts will be published in respective Conference Series International Journals.
Abstracts will be provided with Digital Object Identifier by